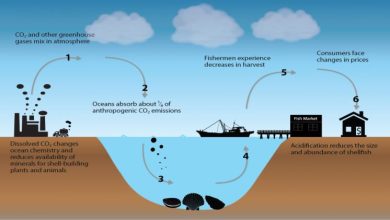
Acid Rain – Causes and Benefits
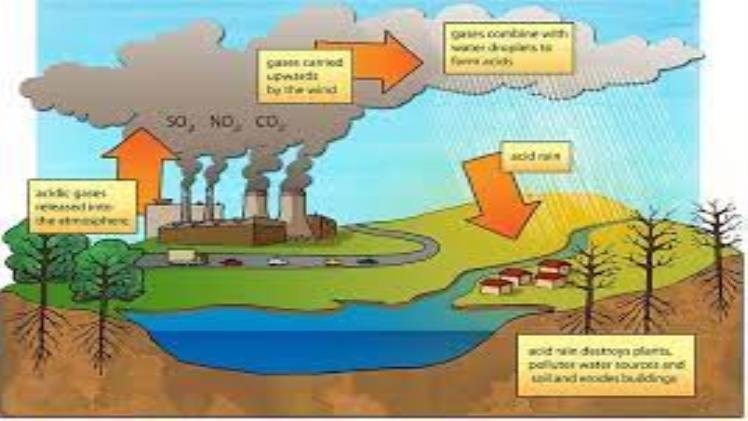
What is Acid Rain? Acid Rain is a phenomenon in which precipitation becomes unusually acidic. Most water has a pH level between 6.5 and 8.5. Acid rain is caused by the excessive release of hydrogen ions into the air, and this is a problem for our environment. There are ways to mitigate the problem of acid rain, however. Read on to learn more about the causes and benefits of acid rain. We’ve all heard about its dangers.
It is believed that the sulphur from Atikokan is insignificant compared to the levels in the boundary waters. However, scientists were able to study the net international flow of emissions and discovered that sulphur from the Ohio Valley, Pennsylvania, and New England were the main contributors to acid rain in the US. As a result, the study led to new legislation that aims to reduce acid rain. But there are still many questions.
One of the most effective ways to reduce acid rain is to reduce energy consumption in general. The less energy you use, the less pollution power plants emit. Another solution is to add powdered limestone to lakes. This process, called liming, is costly and must be continued for a long time to see results. Nevertheless, some countries, such as Norway and Sweden, have successfully used liming to restore lakes in their areas. Meanwhile, a major liming programme is currently underway in Wales.